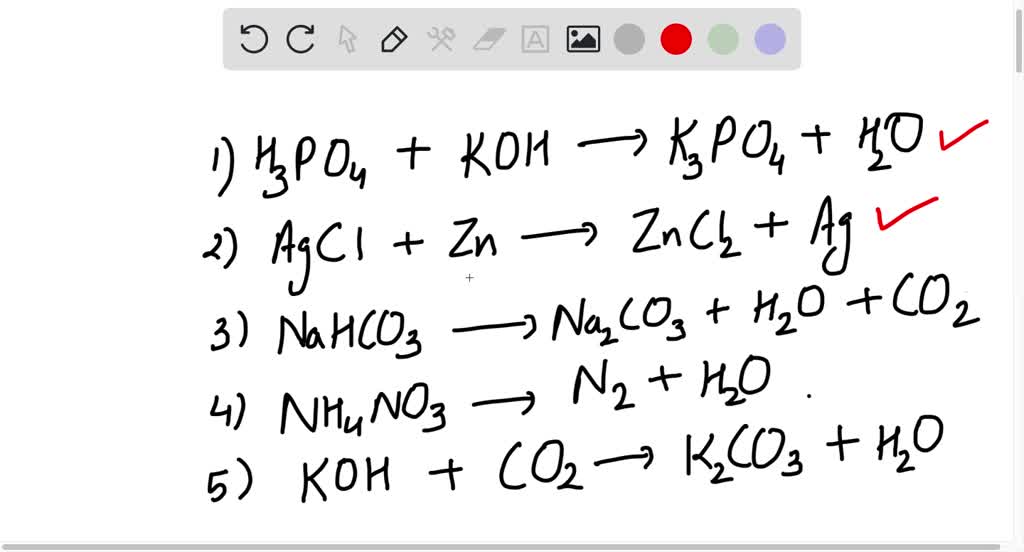
Potassium Hydroxide And Zinc Nitrate . First, we write down the reactants which are zinc nitrate and potassium hydroxide.how many grams of zinc(ii) nitrate and sodium sulfide were consumed to produce this quantity of product?
from www.numerade.com
Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium hydroxide (koh)? The chemical formula for zinc nitrate is.potassium phosphate and calcium chloride;
SOLVED Write an unbalanced equation to represent each of the following
Potassium Hydroxide And Zinc Nitrate What was the concentration of each ion in the original. Write a balanced molecular equation and net ionic equation for the reaction of zinc nitrate and potassium hydroxide. the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and.© 2024 google llc.
From pediaa.com
Difference Between Potassium Hydroxide and Sodium Hydroxide Potassium Hydroxide And Zinc Nitrate Zn (no 3) 2 + 2 koh → zn (oh) 2 + 2 kno 3. What was the concentration of each ion in the original. First, we write down the reactants which are zinc nitrate and potassium hydroxide. the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of. Potassium Hydroxide And Zinc Nitrate.
From pediaa.com
Difference Between Potassium Hydroxide and Sodium Hydroxide Potassium Hydroxide And Zinc Nitrate the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and. Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium hydroxide (koh)? Answer a 2 k 3 po 4 (aq) + 3 cacl. Potassium Hydroxide And Zinc Nitrate.
From www.youtube.com
Does Potassium hydroxide (KOH) and Zinc sulfate (ZnSO4) form a Potassium Hydroxide And Zinc Nitrate the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and.© 2024 google llc.zinc + copper nitrate → zinc nitrate + copper. Zinc nitrate and sodium carbonate; Write a balanced molecular equation and net ionic equation for the. Potassium Hydroxide And Zinc Nitrate.
From www.numerade.com
SOLVED Write an unbalanced equation to represent each of the following Potassium Hydroxide And Zinc Nitrate The chemical formula for zinc nitrate is. Zn (no 3) 2 + 2 koh → zn (oh) 2 + 2 kno 3. the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and. Answer a 2 k 3 po 4 (aq) +. Potassium Hydroxide And Zinc Nitrate.
From www.chegg.com
Solved 17. Iron(II) nitrate [Fe(NO3)2 (aq)] reacts with Potassium Hydroxide And Zinc Nitrate In this type of reaction a more reactive metal displaces a less reactive metal from its salt.zinc + copper nitrate → zinc nitrate + copper. What was the concentration of each ion in the original. Zinc nitrate and sodium carbonate; Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium. Potassium Hydroxide And Zinc Nitrate.
From www.slideshare.net
Precipitation react2 Potassium Hydroxide And Zinc Nitrate What was the concentration of each ion in the original.zinc + copper nitrate → zinc nitrate + copper. Write a balanced molecular equation and net ionic equation for the reaction of zinc nitrate and potassium hydroxide. Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4) 2 (s) + 6. Potassium Hydroxide And Zinc Nitrate.
From www.sjzxlwhg.com
Potassium Hydroxide Solution Potassium Hydroxide And Zinc Nitrate Zinc nitrate and sodium carbonate; In this type of reaction a more reactive metal displaces a less reactive metal from its salt.© 2024 google llc. First, we write down the reactants which are zinc nitrate and potassium hydroxide. Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4) 2 (s). Potassium Hydroxide And Zinc Nitrate.
From www.toppr.com
Identify the type of reactions taking place in each of the following Potassium Hydroxide And Zinc Nitratepotassium phosphate and calcium chloride;© 2024 google llc. Zinc nitrate and sodium carbonate; the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and. Write a balanced molecular equation and net ionic equation for the reaction of zinc nitrate. Potassium Hydroxide And Zinc Nitrate.
From hcs-lab.com
Potassium hydroxide, 0.5N in ethanol HCS Scientific & Chemical Pte Ltd Potassium Hydroxide And Zinc Nitrate Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4) 2 (s) + 6 kcl (aq) answer b zn(no 3) 2 (aq) + na 2 co 3 (aq) → znco 3 (s) + 2 nano 3 (aq) First, we write down the reactants which are zinc nitrate and potassium hydroxide. Zn (no. Potassium Hydroxide And Zinc Nitrate.
From melscience.com
Reactions of potassium and potassium hydroxide MEL Chemistry Potassium Hydroxide And Zinc Nitrate Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium hydroxide (koh)? Zn (no 3) 2 + 2 koh → zn (oh) 2 + 2 kno 3. First, we write down the reactants which are zinc nitrate and potassium hydroxide. Write a balanced molecular equation and net ionic equation for the reaction. Potassium Hydroxide And Zinc Nitrate.
From unitradecapital.com
Potassium Hydroxide 90 unitradecapital Potassium Hydroxide And Zinc Nitrate What was the concentration of each ion in the original. the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and.zinc + copper nitrate → zinc nitrate + copper.potassium phosphate and calcium chloride; Zn (no 3) 2 +. Potassium Hydroxide And Zinc Nitrate.
From lauriekyrran.blogspot.com
Potassium Hydroxide And Iron Ii Nitrate Precipitate LaurieKyrran Potassium Hydroxide And Zinc Nitrate Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4) 2 (s) + 6 kcl (aq) answer b zn(no 3) 2 (aq) + na 2 co 3 (aq) → znco 3 (s) + 2 nano 3 (aq) First, we write down the reactants which are zinc nitrate and potassium hydroxide. Zn (no. Potassium Hydroxide And Zinc Nitrate.
From sea-hill.co.th
105033 Potassium hydroxide seahill.co.th Potassium Hydroxide And Zinc Nitrate Zinc nitrate and sodium carbonate; Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium hydroxide (koh)? the synthesis of zno nanoparticles study from zinc nitrate (zn (no 3) 2) precursor under the three precipitator solutions consists of potassium hydroxide (koh), sodium hydroxide (naoh), and. Zn (no 3) 2 + 2. Potassium Hydroxide And Zinc Nitrate.
From www.youtube.com
Equation for NaNO3 + H2O (Sodium nitrate + Water) YouTube Potassium Hydroxide And Zinc Nitrate In this type of reaction a more reactive metal displaces a less reactive metal from its salt.how many grams of zinc(ii) nitrate and sodium sulfide were consumed to produce this quantity of product? The chemical formula for zinc nitrate is. Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4). Potassium Hydroxide And Zinc Nitrate.
From www.chemicals.co.uk
Potassium Hydroxide Uses, Formula, Dangers & More Potassium Hydroxide And Zinc Nitratezinc + copper nitrate → zinc nitrate + copper. Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4) 2 (s) + 6 kcl (aq) answer b zn(no 3) 2 (aq) + na 2 co 3 (aq) → znco 3 (s) + 2 nano 3 (aq) What was the concentration of. Potassium Hydroxide And Zinc Nitrate.
From www.jiahengyuanchem.com
POTASSIUM HYDROXIDE factory and manufacturers Jiahengyuan Potassium Hydroxide And Zinc Nitrate First, we write down the reactants which are zinc nitrate and potassium hydroxide. Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium hydroxide (koh)? In this type of reaction a more reactive metal displaces a less reactive metal from its salt. the synthesis of zno nanoparticles study from zinc nitrate. Potassium Hydroxide And Zinc Nitrate.
From ramonnewscook.blogspot.com
Zinc Nitrate + Sodium Carbonate Balanced Equation Potassium Hydroxide And Zinc Nitrate Objectivezn (no3)2 + koh = zn (oh)2 + kno3what happens when zinc nitrate (zn (no3)2) react with potassium hydroxide (koh)? The chemical formula for zinc nitrate is. Answer a 2 k 3 po 4 (aq) + 3 cacl 2 (aq) → ca 3 (po 4) 2 (s) + 6 kcl (aq) answer b zn(no 3) 2 (aq) + na 2. Potassium Hydroxide And Zinc Nitrate.
From www.chegg.com
Solved Complete the table below by deciding whether a Potassium Hydroxide And Zinc Nitrate The chemical formula for zinc nitrate is. First, we write down the reactants which are zinc nitrate and potassium hydroxide.potassium phosphate and calcium chloride; Write a balanced molecular equation and net ionic equation for the reaction of zinc nitrate and potassium hydroxide.how many grams of zinc(ii) nitrate and sodium sulfide were consumed to produce this quantity. Potassium Hydroxide And Zinc Nitrate.